
ARTICLE HIGHLIGHTS
- Your digestive tract breaks down carbohydrates and produces glucose. Glucose is a primary source of chemical energy in the human body. Insulin, secreted by the pancreas, helps shuttle glucose from the blood into cells for energy or storage.
- Large blood sugar spikes (often from high-carb foods) cause repeated insulin surges. Eventually, cells can become less sensitive to the hormone, requiring your body to produce more and more. This is called insulin resistance.
- Eventually, your body can’t make enough insulin to keep up with glucose, and your blood sugar stays chronically high. This is prediabetes or Type 2 diabetes and can lead to health problems like heart disease, cognitive decline, reproductive issues, cancer, and more.
- There isn’t a scientific consensus on optimal insulin levels. However, summarized comments from five Levels advisors suggest that fasting insulin should stay below 10 μU/mL.
- Fasting insulin is not typically included as part of routine blood work, partly because some still don’t see insulin as a precursor to other health problems and partly because of inconsistency in testing.
- TAKEAWAY: Insulin is a hormone that affects every cell in your body. Hyperinsulinemia (chronically high insulin levels) and insulin resistance can lead to Type 2 diabetes and a host of symptoms and other conditions. Keeping insulin low is beneficial to your health. Although doctors don’t typically measure insulin, you can still get your levels tested.
A core tenet of metabolic health—the way our bodies make and use energy—is that we require insulin to process blood glucose (or “blood sugar”). It’s the hormone that helps shuttle glucose from the blood into cells. But that’s just one of countless roles played by this essential hormone.
Literally every organ in your body has a sensitive relationship with insulin, and because of that, insulin dysfunction—including insulin resistance—is a root or contributing cause to several chronic conditions, such as heart disease, diabetes, infertility, Alzheimer’s disease, and more. And unfortunately insulin resistance affects more than 133 million Americans, and potentially up to 88% of US adults. So you’d think this vital hormone would be something we test and monitor frequently, but it is not—for a number of reasons. Here, we’ll explore what insulin is, how it impacts your health, why it’s difficult to determine “normal” insulin levels, what insulin levels you should aim for, and how to get tested.
What is insulin?
Insulin is a hormone—a chemical “messenger” that circulates through your bloodstream and triggers physiological events. Produced in your pancreas (a small organ that sits behind your stomach) by specialized β-cells (pronounced “beta cells”), insulin affects every single cell in your body, including your bone, brain, muscle, and skin cells. Of course, given the variety of purposes of these (and other) cells, it’s little surprise that insulin has some distinct actions across these tissues.
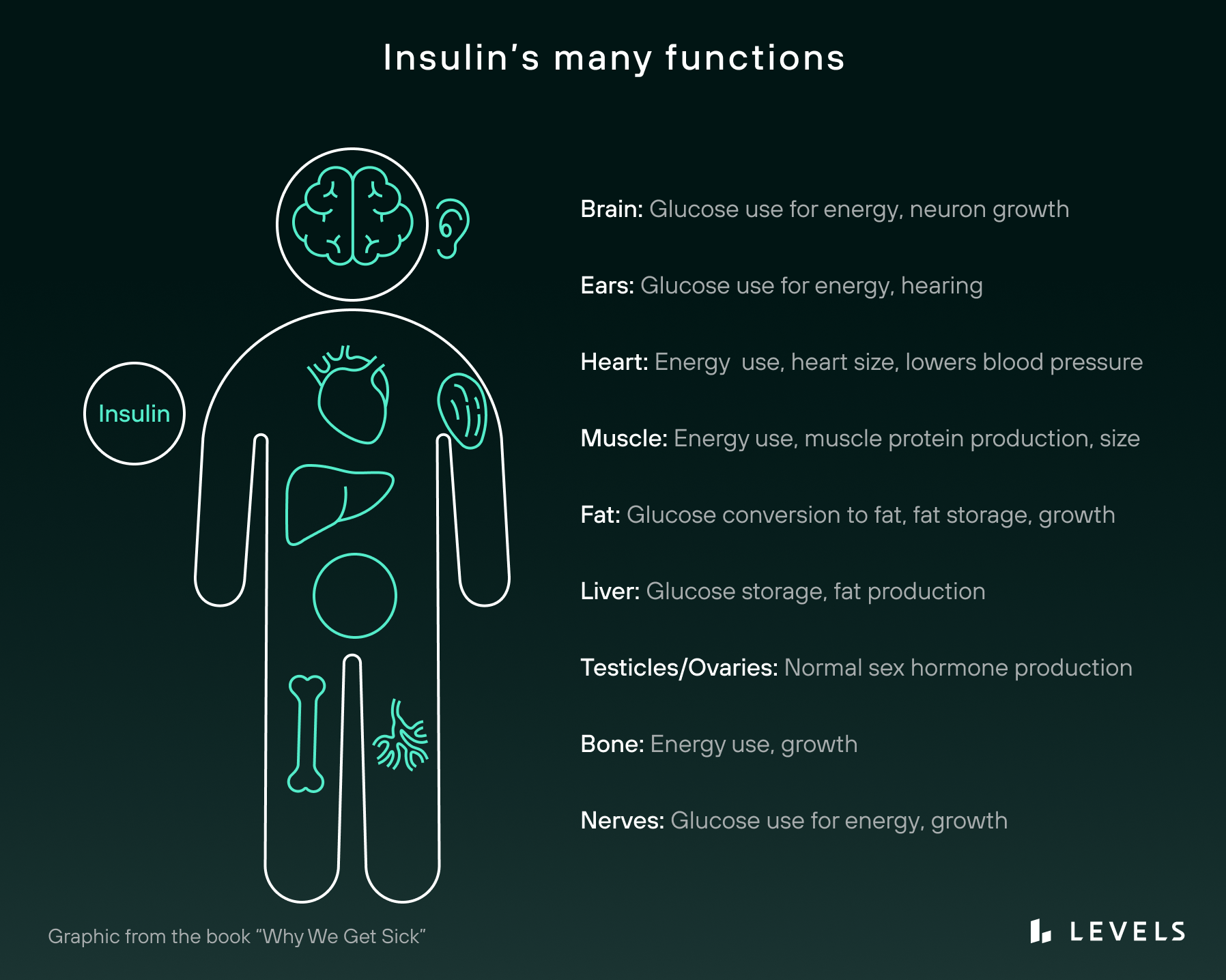
As an anabolic hormone, insulin promotes growth in bones, skeletal muscles, and other tissues. In the brain, there is evidence that it governs cognition, neuronal plasticity, and memory processing. In the kidneys, it helps regulate sodium absorption and homeostasis. And in the liver, insulin helps synthesize alternative energy sources, such as glycogen and fatty acids.
But perhaps the most important function insulin serves is the one it’s best known for: helping your body extract chemical energy from food. When you eat a meal, your digestive tract breaks down carbohydrates and produces, among other things, a simple sugar called glucose. As your blood glucose rises above its baseline, or homeostasis level, your pancreas responds by secreting insulin to help you metabolize this glucose by shuttling it into cells for energy or into storage. This temporarily raises your level of insulin. When the excess glucose has been accounted for, your insulin level falls back to its baseline again.
Insulin and glucose levels are tightly linked in this way because both are necessary to fuel your body on the cellular level. Glucose itself is a main source of chemical energy in the human body, while insulin is the key that unlocks many cells, allowing glucose to enter.
For example, consider a muscle cell. When insulin in your bloodstream comes into contact with that cell, it latches onto the cell’s insulin receptor. This sparks a chemical reaction that brings a special protein to the cell’s surface. This protein is called GLUT4, aka “Glucose Transporter Type 4.” The GLUT4 protein then grabs onto a glucose molecule from your bloodstream and shuttles it back into the cell. There, the cell breaks down the glucose into a more refined form of chemical energy called ATP.
Note that GLUT4 is only one of many glucose transporter proteins that have been discovered—and glucose is not the only energy source that cells can tap into. Even so, the process described above is the dominant way cells obtain chemical energy in the human body. When this process breaks down, the body becomes metabolically unstable with dangerous health consequences.
Due to insulin being overlooked for so long, there is no scientific consensus on optimal insulin levels.
Even in tissues that don’t require insulin to open the cellular doors for glucose, the cells still rely on insulin to tell the cell what to do with the glucose (or any other energy). Unlike the muscle, the liver doesn’t need an insulin signal to pull in glucose from the blood, but that insulin signal is essential to promote the storage of the glucose as glycogen, or even converting the glucose into fats.
Across the muscle, liver, and other tissues, one well-known example of this type of dysfunction is diabetes mellitus. Diabetes is fundamentally a problem of insulin regulation. People with Type 1 diabetes cannot create enough insulin naturally to get glucose into their cells. Without treatment, they may quickly enter a health crisis. Type 2 diabetes, on the other hand, often features excess insulin that becomes less effective. When the body doesn’t respond to insulin properly, glucose levels remain uncontrolled even though insulin levels are chronically elevated. This cascades into many other serious health issues.
Learn more:
Why Do People Without Diabetes Care about Insulin?
Diabetes is perhaps the most prominent insulin-related illness, affecting 37.3 million people (11.3% of the population) in the US. But it is by no means the only one. Many other serious health conditions have been linked to chronically high insulin (hyperinsulinemia) and its close counterpart: insulin resistance.
“Insulin resistance” simply means your cells have stopped responding appropriately to insulin. There are several mechanisms that can drive this condition, including genetics, overnutrition, excess fructose, and excess visceral fat accumulation. Another important one is the chronically elevated insulin, which can result from frequent refined carb consumption.
In the latter case, high glycemic variability and chronically high blood glucose levels (hyperglycemia) cause repeated surges of insulin. As cells are exposed to more and more of the hormone, they become less and less sensitive to it. This “numbing” effect can impact you in diverse ways because almost every cell in your body has insulin receptors. And it can take hold surprisingly quickly. In one study, for instance, researchers forced insulin levels to stay … Read the rest










